Introducing the DECTRIS PILATUS4 CdTe 1M Detector at the Swiss Light Source

A 5-minute read
Discover the MX group members’ journey of integrating a DECTRIS PILATUS4 detector into their beamline control system at the Swiss Light Source (SLS). Learn about the remarkably smooth integration, minimal effort, and great user experience!
The prototype of a DECTRIS PILATUS4 CdTe 1M detector was tested at the SLS PXIII beamline for protein crystallography. The aim was assessing the detector’s suitability for the beamline after the upgrade of the SLS synchrotron to SLS 2.0, which just started. The final two weeks of SLS’ operation could be used efficiently for testing, and even for collecting datasets - also made possible through the fast and smooth integration of the detector.
We asked members of the SLS’ Macromolecular Crystallography (MX) group about their experience.
Dr. Meitian Wang, Head of MX
Wayne Glettig, Group Leader for MX Instrumentation
Dr. Vincent Olieric, Beamline Scientist in the MX Applications group
Dr. Kate Mary Louise Smith, Software Developer in the MX Data group
Dr. Ezequiel Panepucci, Software Developer in the MX Data group
DECTRIS: How did you integrate PILATUS4 into your beamline control system?
Ezequiel: Integrating the PILATUS4 detector into our beamline control system was a straightforward process, due to the fact that the platform came equipped with the same SIMPLON interface we use to control our EIGER detectors (the data and control interface SIMPLON API). This allowed us to leverage our existing Python modules to seamlessly control the PILATUS4 detector.
The integration was remarkably easy, with us being able to configure the detector and initiate a data acquisition series within a minute of its being online on the network. From my experience, having worked on integrating various detectors, this was one of the smoothest integrations we've ever conducted.
Wayne: We initially anticipated that it would require a significant amount of effort. However, to our pleasant surprise, the process went exceptionally smoothly and was executed efficiently.
Ezequiel: Another notable aspect was the immediate availability of streamed images for our data analysis software when conducting grid scans. This was a significant advantage, as it spared us the need to make any adjustments to our existing software infrastructure. The data generated by the PILATUS4 detector were seamlessly compatible with our analysis tools, making the entire process highly efficient and user-friendly. It was definitely a major advantage in terms of data accessibility and compatibility.
Could you describe the level of effort that was required to get the detector up and running, from the moment it was delivered to the point when you acquired your first scientifically usable dataset?
Ezequiel: If we were to quantify it, I would say it required absolutely minimal effort - effectively zero. That's how smooth the process was. We began the setup in the morning, and right after lunch, the system was essentially up and running.
The most challenging parts of the setup process were identifying the MAC address on our network and dealing with the required 802.1x authentication in our network, which took approximately 3 minutes.
Did you encounter any particular challenges?
Kate: The process was relatively straightforward, and we didn't face any significant issues.
For automatic data processing, it worked seamlessly 'out of the box' with the X-ray Detector Software. Configuring the detector’s dimensions in the input file was a straightforward process, requiring just a simple click. It was quite a user-friendly experience.
What was the maximum speed at which you collected data with the PILATUS4, and what do you expect in terms of speed once the upgraded SLS 2.0 synchrotron is operational?
Meitian: We were able to achieve data collection speeds of up to 500 Hz with the PILATUS4. In practice, 100-200 Hz is a sufficient rate for daily operations, but we might want to reach a few hundred more Hz for specific projects.
Wayne: With significantly higher flux after the SLS upgrade, we could potentially push the data collection speed even further, aiming for a maximum of 2,000 Hz in extreme scenarios.
What is your assessment of using a pixel size of 150 µm for protein crystallography?
Vincent: Currently, spot separation isn't a significant concern for us, mainly because we're no longer dealing with large Unit Cells that require extensive separation. The spot separation process is usually straightforward when it comes to fragment screening and working with well-characterized proteins. In fact, a significant portion of our crystal samples that are brought to synchrotrons and beamlines fall into this category.
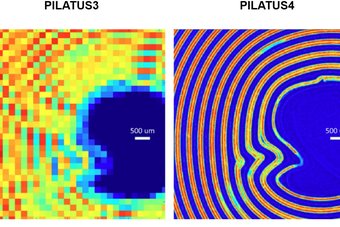
Wayne: One advantage of using larger pixels (compared to those of EIGER) is the reduction in the number of pixels that are required to cover a surface, which in turn means faster data processing pipelines and less storage space. However, it's essential to verify whether similar data quality can be achieved with fewer pixels. This assessment should be based on data quality considerations, independent of the pixel size.
Related to the larger pixel size, can you solve the structure effectively and observe high-resolution details such as ligands?
Vincent: Absolutely. This can even lead to accelerated data processing and more efficient data transfer downstream.
After you’ve utilized the 1M-pixel CdTe detector for your tests, what type of detector would you consider as the optimal choice for your beamline?
Kate: Our preference remains with silicon as the ideal choice because it continues to perform effectively, even at lower energies . We still require the capability to collect high-quality data at these lower energies, and this makes silicon a compelling option for our needs.
Do you recommend the PILATUS4 detector for Macromolecular Crystallography? And, if so, for which specific applications?
Vincent: It is highly recommended for our conventional crystallography work, such as regular cryo-measurement and fragment screening. It performs exceptionally well in these applications.
With the upgrade to SLS 2.0 and a PILATUS4 detector, what is the maximum resolution you would anticipate achieving for protein crystallography?
Meitian: For protein crystallography, we aim to achieve a resolution of around 1.5Å, which is likely the upper limit. However, when it comes to other applications, such as chemical crystallography, we may target a resolution of approximately 0.7Å. These applications involve using higher X-ray energy and a larger detector, e.g. a PILATUS4 2M.
We thank the MX group members for sharing their expertise and look forward to still more exciting developments at their beamline in the future!
Discover our PILATUS4 product pages
Questions about PILATUS4 or Crystallography? Contact our experts!
Dr. Tilman Donath
Product Manager - High Energy
E. tilman.donath@dectris.com
Dr. Sofia Trampari
Application Scientist Crystallography
E. sofia.trampari@dectris.com